Topic: The states of the matter
Abstract: This learning proposal aims for students to understand the three states of matter: solid, liquid and gas: what this has to do with matter composed by particles (atoms and molecules) with electric bounds and movement. It also relates energy with heat and the transformation of this in particle movement.
Learning Objectives:
- To know or review the concept of matter composed by particles
- To know or review the concepts of atoms and molecules
- To know or review the composition of water molecules and the electric bounds between them
- Relate these concepts with day-to-day life experiences
- Relate these concepts with art expression: To know how different authors of classical music and painting explain the phenomena and changes in nature in order to understand them.
- To dance individually and collectively representing physical phenomena
- To mobilize linguistic, communicative, communicative, organizational and teamwork skills by performing together the concepts to learn.
Teachers’ profile: (team transversality) : Sciences, Arts, Languages
Advisable age of students: (12-13 years) Attention to diversity and inclusion aspects to have in account:
Previous knowledge:
- Previous knowledge about the structure of matter
Global development
Total duration: Approximately 2 hours
Materials:
- Pictorial works showing water in its different states: snowy, ice landscapes, liquid (ocean, rivers), gaseous (smoke, clouds)
- Ice cubes, containers, heater, and pan to meld the ice and to boil the water
- Music and music equipment to play in the classroom: slow, quiet piece, faster harmonic piece, very vigorous piece.
- Cardboard, string and pen markers to label the particles H20 or the atoms H, O
Phase 1 30 min
Development: Art showing different states of matter:
• Solid (snow, icy landscapes)
• Liquid (oceans, rivers)
• Gaseous (smoke, clouds)
Can we associate these states with movement?
In which state do you see more movement?
Match state with movement and this with music
Phase 2 60 min Artistic action and experimentation
Development:
1- Share and collect previous knowledge. Conversation and debate between the children about what do they know:
Are the pictures we saw different type of matter? why can the same kind of matter have different states?
2- In groups of 4 or 5 students explore and look for answers to the previous questions and check their hypothesis
3- Put the investigated facts together:
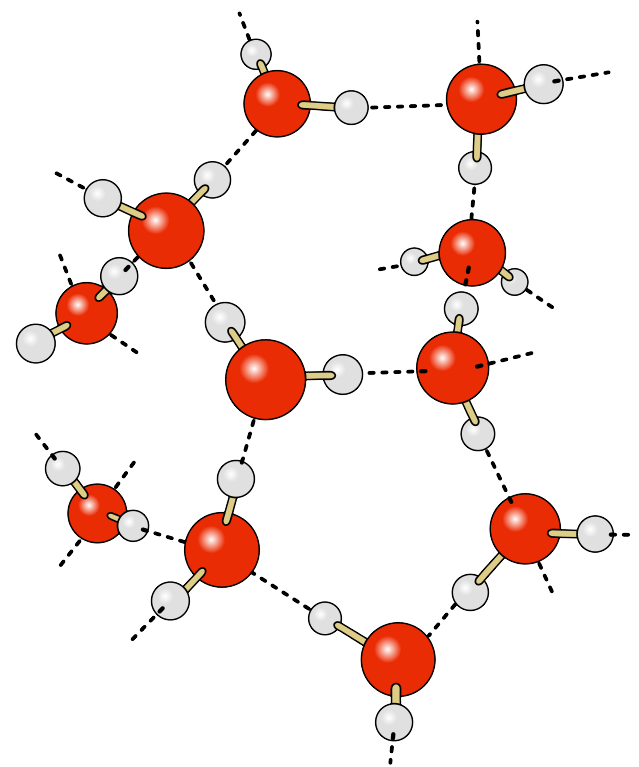
• Talk about composition of matter: For example, Water is composed by H20 molecules (which is formed by three atoms: 2 Hydrogens, 1 Oxygen)
• How do the molecules stick together?
• How can we turn solid water in liquid water and liquid water in gaseous water?Different behavior of a solid, a liquid and a gas in relation with the space they occupy/fill. This can be done in a practical way: Having ice cubes in the class and talking about the space they occupy: do they need a container? Melt down the ice cubes: does the liquid need a container. How much does it occupy? Boil the liquid to the gas: Where does it go?
4- It will come out that:
• Water molecules stick together because of the electric forces bounds that exists among them.
• All the molecules are moving (vibration movement and other movements).
• If we provide of energy (heat) they absorb this energy and move faster.
• If they move faster, with more energy, they break some of the bonds and expand into the space (liquid)
• If we provide even more energy, they move so fast that they totally break the bounds and each molecule moves freely around the whole space they have
5- Theater action:
• The students are going to be the water particles. A molecule can be represented either by a student or by 3 students holding arms together (H2O -Two Hydrogens and 1 Oxygen).
• The teacher will have prepared 3 kinds of music:
– Slow music to represent low energy – cold (ice-solid state)
– Faster melodic music to represent medium energy – temperate (liquid state)
– Very vigorous music to represent high energy – hot (gaseous state)
• The students represent the state of the matter grouping and dancing according to the music the teacher plays:
– At the slow music: They will stick together, making bounds with their arms, occupying a small space, and moving slowly all together at the same time. They will represent solid state, because is cold.
– At the faster melodic music: They get some more energy. They will move harmonically around the space, in a flow way, moving together but occupying more space: They will represent liquid state, it’s warm
– At the very vigorous music: Every particle (student or group of 3 students) will move freely and fast around all the space, bouncing on the walls and occupying all of it. They will represent gaseous state. It’s hot.
Phase 3: 20 min Reflexion and debate
Development:
The students sit down again in rounds and talk about the experience and what they understand about it, reviewing the concepts of energy, related to movement, and the concept of the structure of matter composed by particles which are atoms and atoms forming molecules.
References and links:
1- Ice, water, and gas pictures:



Claude Monet
2- Music to play solid, liquid and gas: Music teacher can help with that
Solid: Winter music
Liquid: Johan Strauss – Danube blue waltz
Gas: https://youtu.be/fKC-LU669r8?t=167
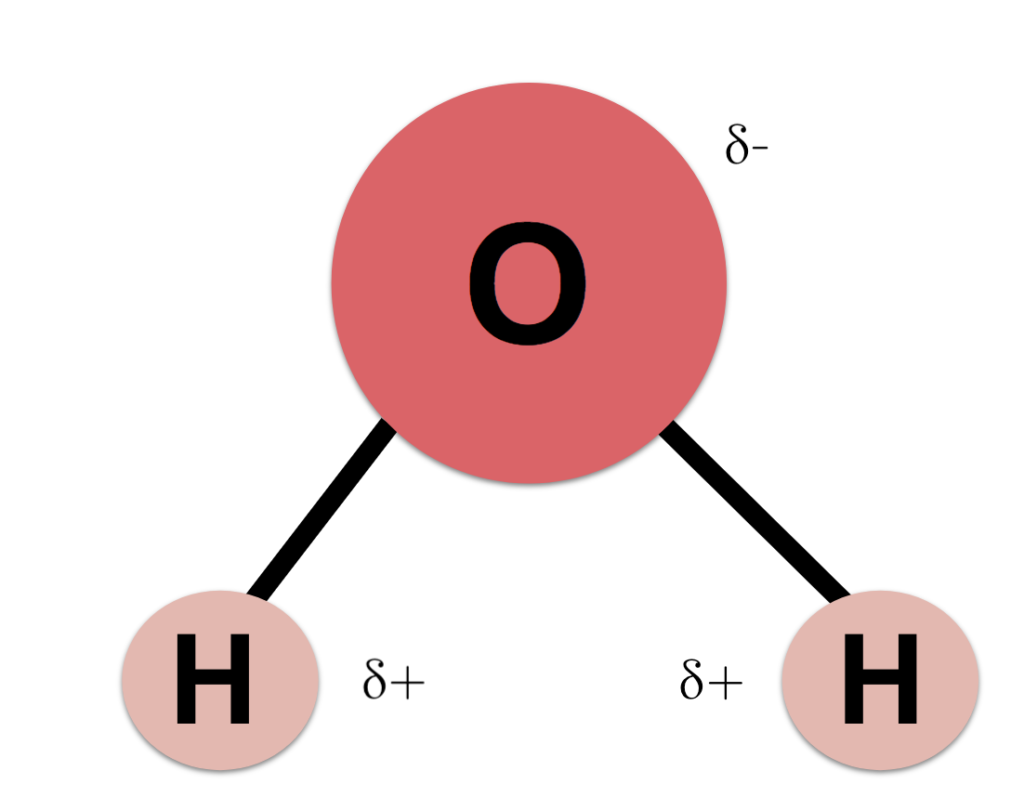
3. One water molecule
Several water molecules holding together by electrical bounds. Every molecule moves and vibrates. If we provide energy this movement increases and eventually the electrical bounds break, we go from solid to liquid state and, when every molecule is totally free, to gaseous state.
4- Chemical party: Video with human performances about the behavior of atoms and molecules:

